Generic vs brand name drugs? Generic and brand name drugs both are safe, effective, and bioequivalent. In the United States of America, 9 out of 10 prescriptions filled are for generic drugs [1]. But there are some differences between generic and brand name drugs. Most of the time we use the terms generic and brand name drugs and generic and brand name medicine interchangeably. Generally, the term “generic and brand name of drugs” and “generic medicine and brand medicine” both are used as the same meaning to understand a pharmaceutical product. However, do you know the difference between Drug and Medicine? After reading this article you will get the answer to the following questions:
- Firstly the difference between generic and brand name drugs.
- Secondly the difference between generic and patent drugs.
- Thirdly the difference between generic medicine and patent medicine.
- Fourthly the difference between generic and original drugs.
- Fifthly Difference between generic medicine and brand medicine.
Table of Contents
Generic vs Brand Name Drugs
Before reading this article you should read Generic name of drugs, Brand name of Drugs, Generic drug/ medicine, Brand name drug/medicine to clear the concept.
Definition of generic vs brand name drugs
A generic drug is an off-patent pharmaceutical product that is manufactured by a pharmaceutical company in the same strength, dosage form, route of administration, safety, quality, performance characteristics, and intended use after expiring the patent of relevant brand name drug (Innovator drug).
According to the World Health Organization (WHO), “generic drug” or “generic medicine” is a pharmaceutical product which is:
–interchangeable with an innovator product,
–manufactured without innovator company license, and
–marketed after the patent or other exclusive rights expiration [2].
According to USFDA (United States Food and Drug Administration) defines a generic drug as:
‘A drug product that is comparable to a brand/ RLD (reference listed drug) product in strength, dosage form, route of administration, performance characteristics, quality, and intended use’ [3].
According to EMA (European Medicines Agency), ‘A generic medicine is a medicine that is developed to be the same as a medicine that has already been authorized (reference medicine). Generic medicine contains the same active ingredient(s) and it is used at the same dose(s) as the reference medicine. However, the name of the medicine, its appearance (for example color or shape), and its packaging can be different from those of the reference medicine’ [4].
On the other hand, a Brand name drug is a pharmaceutical product that is developed and marketed under a patent or registered trademark by a pharmaceutical company. But it is approved after establishing the drug’s safety and effectiveness through animal and clinical (human) studies. Also, brand name drugs known as innovator drugs.
Frequently, a drug can be developed in several ways such as buffered versus non-buffered aspirin, resulting in multiple brand names for the same drug (Active Pharmaceutical Ingredient).
Patents and Exclusivities
A Generic drug is an off-patent pharmaceutical product that may manufacture by various companies. While A brand name drug is patent protected that disallow other company for selling generic versions of the same drug.
Trade Name
A generic drug is marketed under the generic name of the drug. On the other hand, a brand name drug is marketed under a unique, proprietary name given by a pharmaceutical company.
Application for USFDA approval
Abbreviated New Drug Application (ANDA) is required for USFDA approval for marketing authorization. On the other hand, New Drug Application (NDA) is required for USFDA approval for marketing authorization.
Manufactured by
A generic drug is manufactured by several pharmaceutical companies after the expiration of patents and marketing exclusivities of the relevant brand name drug. While a brand name drug is developed and manufactured by an innovator company.
Animal & Clinical (Human) study for generic vs brand name drugs
Since a brand name drug is established after clinical study and generic drug is a replica of brand name drug that is why clinical study not required to perform. While animal and clinical (human) studies are essential to prove the safety and effectiveness of a new brand name drug.
Price of generic vs brand name drugs
A generic drug is cheaper than its brand name drug. Generic drugs are 85% less than the brand-name drugs [5]. Whereas a brand name drug is costly. According to the Congressional Budget Office, generic drugs save consumers approximately 8-10 billion dollars per year at retail pharmacies [6]. According to the IMS Health Institute, in the United States healthcare system, generic drugs saved 1.67 trillion dollars from 2007 to 2016 [7]. Generic medicines are typically 20 to 90% cheaper than brand name drugs [8].
Why generic drugs are cheaper?
- Animal and clinical (human) studies not essential for generic drug applicants to establish the safety and effectiveness of that generic drug.
- Lessen time and money required in drug product research and development because Innovator Company already discloses the excipients (Inactive ingredients) of that brand name drug.
- No promotional budget required.
- Does not required to buy the right to sell the generic version of the brand name drug.
- After the expiration of a brand name drug, market competition between multiple generic companies typically results in a low price.
Appearance (Color, Shape, Size) of Generic vs brand-name drugs
A generic drug may look different in shape, size, and color from relevant brand name drug. While a brand name drug has a unique look as design during product development.
In the United States, trademark laws do not allow a generic drug to look exactly identical to other existing drugs on the market. A generic drug may be different from the brand name drug in:
- Shape,
- Size,
- Color, and
- Packaging. Read: Packing vs Packaging
Name variation
Generally, Generic drug has the same name as its generic name of the drug (API) in any country. For example, Paracetamol tablet is a generic drug. While the name of a brand-name drug may same or different in different countries. For example, Panodil is the brand name of Paracetamol in Denmark, Iceland, Norway, Sweden. Moreover, Daleron, Depon, Dexamol, Dolex, Dolo, Para, NAPA tablet are the popular brand name drug of Paracetamol.
Excipients in generic vs brand name drugs
A generic drug may contain the same or altered excipients (inactive pharmaceutical ingredients) from relevant brand name drug. While a new brand name drug may contain necessary acceptable excipients by innovator company during development.
Availability
A generic drug is available on the market after the expiration of periods of patent and marketing exclusivity. While a brand name drug is available on the market after approval from the FDA by proving the safety and effectiveness of a new brand name drug.
Examples of generic vs brand name drugs
List of generic and brand name drugs
| Name of Generic Drugs | Name of Brand Name Drug |
| Aspirin | Aspirin, Aspergum, Aspir-Low, Bufferin, Ecotrin, Empirin, Sloprin, and Zorprin. |
| Paracetamol | Tylenol, Acephen, NAPA, Mapap, Nortemp, Ofirmev, Acamol. Acetalgin, Calpol, Febridol, Hedanol, Herron, Influbene, Lekadol, Pacimol, Panado, Para, and Panadol. |
| Ibuprofen | Advil, Motrin, and Nurofen. |
| Metformin | Glucophage, Fortamet, and Glumetza. |
| Glibenclamide | Glynase, Diabeta, Flycron, Daonil, Euglucon, Glidanil, Gliben-J, Gilemal, Glybovin, Micronase, and Maninil. |
| Tramadol | Ultram, and Zytram. |
| Favipiravir | Avigan. |
In short, Generic vs Brand name drugs
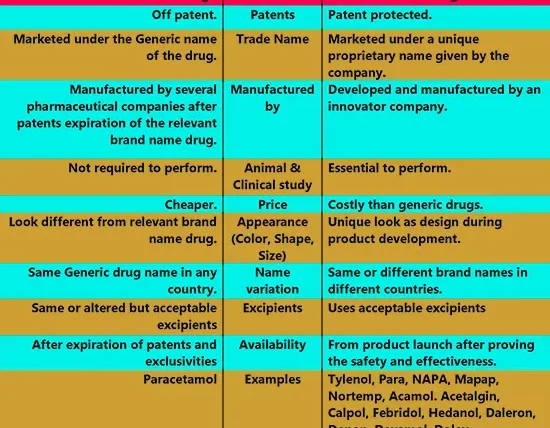
| Features | Generic Drugs | Brand Name Drugs |
| Definition | A generic drug is an off-patent pharmaceutical product that is manufactured by a pharmaceutical company in the same strength, dosage form, route of administration, safety, quality, performance characteristics, and intended use after expiring the patent of the relevant brand name drug (Innovator drug). | A Brand name drug is a pharmaceutical product that is developed and marketed under a patent or registered trademark by a pharmaceutical company. But it is approved after establishing the drug’s safety and effectiveness through animal and clinical (human) studies. Also, brand name drugs known as innovator drugs. |
| Patents | Off patent. | Patent protected. |
| Trade Name | Marketed under the Generic name of the drug. | Marketed under a unique proprietary name given by the company. |
| Application | ANDA required for USFDA approval. | NDA required for USFDA approval. |
| Manufactured by | Manufactured by several pharmaceutical companies after patents expiration of the relevant brand name drug. | Developed and manufactured by an innovator company. |
| Animal &
Clinical study | Not required to perform. | Essential to perform. |
| Price | Cheaper. | Costly than generic drugs. |
| Appearance (Color, Shape, Size) | Look different from relevant brand name drug. | Unique look as design during product development. |
| Name variation | Same Generic drug name in any country. | Same or different brand names in different countries. |
| Excipients | May contain the same or altered but acceptable excipients from relevant brand name drug. | Uses acceptable excipients by the innovator company during development. |
| Availability | After expiration of patents and exclusivities | From product launch after proving the safety and effectiveness. |
| Examples | Paracetamol tablet | Tylenol, Para, NAPA, Mapap, Nortemp, Ofirmev, Acamol. Acetalgin, Calpol, Febridol, Hedanol, Daleron, Depon, Dexamol, Dolex, Dolo, Herron, Lekadol, Pacimol, Panado, and Panadol tablet. |
Image of the difference between generic vs brand name drugs
Similarities of generic and brand name drugs
A generic drugs/ medicine of a brand name drug must have the same:
- Active ingredient(s),
- Strength,
- Indications,
- Dosage form such as tablet or syrup or suspension or emulsion,
- Route of administration such as oral or IV,
- Exact manufacturing standards as the brand-name drug,
- Safe and effective as brand name drug,
- Label as the brand-name drug.
In conclusion, generally, generic drug and generic medicine are the same things as well as brand name drugs, trade name drug, patent drugs, original medicine, innovator drugs, and patent medicine are the same things. But the terms “generic medicine” and “brand medicine” or “brand name medicine” are appropriate than “generic drug” and “brand name drugs”. Also, you may read the difference between Drug and Medicine.
References
| 1. US Food and Drug Administration. Generic Drugs; 2019. https://www.fda.gov/drugs/buying-using-medicine-safely/generic-drugs ; Access date: May 30, 2020. |
| 2. World Health Organization (WHO). Generic drugs. Geneva: WHO; 2015. |
| 3. US Food and Drug Administration (FDA) Generic drugs. Silver Spring, MD: FDA; 2008. Approval of generic drugs. |
| 4. European Medicines Agency (EMA) Questions and answers on generic medicines. Canary Wharf: EMA; 2011. |
| 5. US Food and Drug Administration (FDA). Generic Drug Facts. https://www.fda.gov/media/107601/download ; Access date: 29 may, 2020. |
| 6. Grabowski HG. Chapter 8: competition between generic and branded drugs. Pharmaceutical innovation: incentives, competition, and cost-benefit analysis in international perspective. 2007, p. 153–288. |
| 7. IMS Institute for Healthcare Informatics. The use of medicines in the United States: review of 2011. Danbury (CT): IMS Institute for Healthcare Informatics; 2011. |
| 8. Dunne S, Shannon B, Dunne C, Cullen W. A review of the differences and similarities between generic drugs and their originator counterparts, including economic benefits associated with usage of generic medicines, using Ireland as a case study. BMC Pharmacol Toxicol. 2013;14:1. |