Enzyme, Coenzyme, Apoenzyme, Holoenzyme, and Cofactor are most often confusing. If you want to know about the Enzyme clearly, you must know Coenzyme, Apoenzyme, Holoenzyme, and Cofactor. Now, Enzyme, Coenzyme, Apoenzyme, Holoenzyme, and Cofactor are discussed step by step.
Table of Contents
Enzyme
Enzymes are macromolecular, proteinous, chemical catalysts which are secreted by the exocrine glands that accelerate or catalyze all biochemical reactions in the living body. They are carried by the specific duct and generally functioned near to the glands. Wilhelm Kühne (German physiologist) first used the term enzyme in 1877.
Properties of Enzymes
- Protein in nature except for ribozyme
- Enzymes are colorless but some are blue, green, or greenish brown.
- Colloidal and heat liable
- Soluble in water
- They have definite isoelectric pH
- Don’t initiate or alter the existence of reaction
- Highly specific in reaction
- Remain chemically unchanged in the overall process
- Functionally independent of parent cell
- Inhibitor or activator modify their rate of activity.
Examples of Enzyme
| Cellulases | Papain |
| Ligninases | Rennin |
| Proteases | Lipases |
| Amylases | Trypsin |
| Mannanases | Pectinases |
| Glucanases | Nucleases |
| Betaglucanases | DNA Ligase |
| Amyloglucosidase | Polymerases |
| Pullulanases | Xylanases |
| Acetolactate Decarboxylase (ALDC) | Hemicellulases |
| Lignin Peroxidases |
Apoenzyme
Apoenzymes are those enzymes which require an organic or inorganic cofactor to perform their specific functions but do not have one bound. Apoenzymes also call apoproteins.
In other words, Apoenzymes are enzymes that lack their necessary cofactor(s) for performing their specific function. Apoenzyme is an inactive form of some enzyme. Apoenzyme is also called a proenzyme or zymogen.
Holoenzyme
The Holoenzyme is the combination Apoenzyme & Cofactor that activated complex of an enzyme for a specific catalytic action. Holoenzymes are the active form of an apoenzyme. Here co-factor may be inorganic ions or organic or metallorganic (coenzyme).
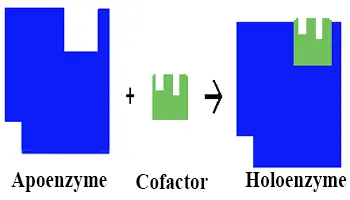
Example of Holoenzyme
- DNA polymerase,
- RNA polymerase etc.
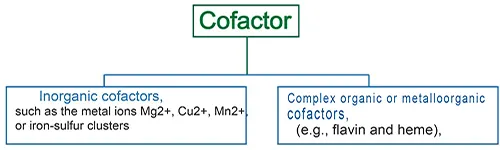
Cofactors
Cofactors are either one or more inorganic (e.g. metal ions and iron-sulfur clusters) or a complex organic or metalloorganic (e.g. flavin and heme), non-protein chemical compounds that assist in the biochemical transformation of an Apoenzyme. Cofactors also known as “helper molecules” that assist apoenzyme during the catalysis of reactions.
Some sources also limit the use of the term “cofactor” to inorganic ions.
Types of cofactor
Cofactors are divided into two broad groups:
- Organic cofactors, such as flavin or heme,
- Inorganic cofactors, such as metal ions Mg2+, Cu2+, Mn2+, or iron-sulfur clusters.
Examples of Inorganic Cofactor
| Enzymes | Cofactors |
| Alkaline phosphate | Zn2+ |
| Hexokinase | Mg2+ |
| Creatine phosphokinase | Mg2+ |
| Cytochrome oxidase | Cu2+/Fe2+ |
Coenzyme
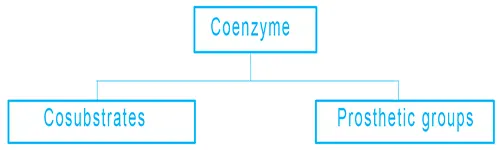
Coenzymes are small, organic or metalloorganic, non-protein molecules that are as auxiliary for the specific action of an enzyme. They act as a transient carrier of specific functional groups from enzyme to enzyme. Coenzymes bind to the apoenzyme and assist in enzyme activity. A coenzyme is technically a type of cofactor.As for Example, NADH, NADPH and adenosine triphosphate (ATP), Riboflavin, Thiamine, and Folic Acid.
These are relatively heat stable low molecular weight compound and highly bound to an enzyme. Most of these are vitamin B-complex derivatives.
| Coenzymes | Derived from Vitamin B-Complex |
| NAD, NADP | Niacin |
| CoASH | Pantothenic acid |
| FAD, FMN | Riboflavin |
| Pyridoxal phosphate | Pyridoxin |
| Thiamine phosphate | Thiamine |
| Tetrahydrofolate | Folic acid |
You may also read:
I don’t leave many remarks, but i did some searching and wound up here Enzyme, Co-enzyme, Apoenzyme, Holoenzyme, and Co-factor. And I actually do have a couple of questions for you if it’s allright. Could it be just me or does it appear like some of these remarks appear as if they are coming from brain dead individuals?:-P And, if you are writing on other online social sites, I’d like to follow anything fresh you have to post.Could you list of every one of your communal pages like your linkedin profile, Facebook page or twitter feed? http://englishdefence-league.co.uk/groups/how-generate-muscle-quickly tricks-for-the-skinny-guy/